Global IBD Treatment Market Forecast 2025–2033: Biologic Innovations Transform Patient Care
Rising prevalence of Crohn’s disease and ulcerative colitis, along with advances in biologic and targeted therapies, is driving steady growth in the inflammatory bowel disease treatment market.
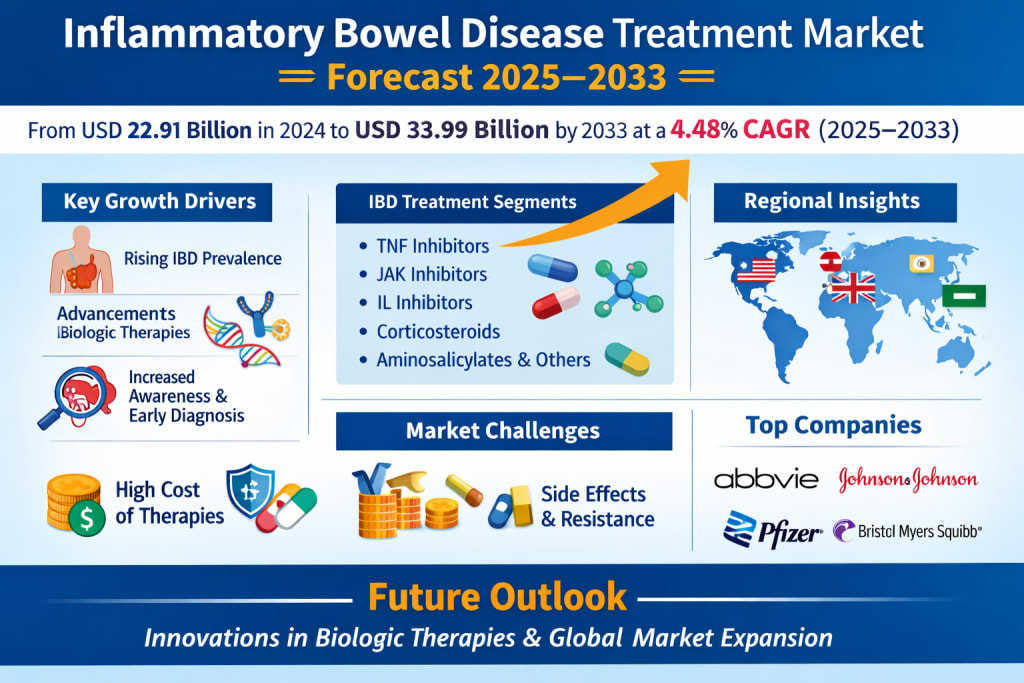
Inflammatory Bowel Disease Treatment Market Forecast and Size 2025–2033
The global healthcare industry continues to experience rapid innovation, particularly in the treatment of chronic diseases. Among these conditions, inflammatory bowel disease (IBD) remains one of the most complex and challenging disorders to manage. With rising diagnosis rates worldwide and major breakthroughs in biologic therapies, the IBD treatment market is steadily expanding.
According to Renub Research, the global inflammatory bowel disease treatment market is expected to grow from approximately USD 22.91 billion in 2024 to nearly USD 33.99 billion by 2033, expanding at a compound annual growth rate (CAGR) of 4.48% between 2025 and 2033. The growth trajectory reflects increasing disease prevalence, expanding access to healthcare, and the development of innovative treatment options.
The market’s evolution is also supported by improving healthcare infrastructure in emerging economies, greater awareness about gastrointestinal diseases, and advances in targeted drug therapies. Together, these factors are reshaping how physicians diagnose and manage inflammatory bowel diseases globally.
Understanding Inflammatory Bowel Disease
Inflammatory bowel disease is a group of chronic conditions that cause persistent inflammation of the gastrointestinal tract. The two most common forms are Crohn’s disease and ulcerative colitis.
Crohn’s disease can affect any part of the digestive tract, from the mouth to the anus, whereas ulcerative colitis primarily targets the colon and rectum. Symptoms often include abdominal pain, severe diarrhea, fatigue, weight loss, and malnutrition. Because these diseases involve an overactive immune response, treatment strategies focus on controlling inflammation and preventing disease flare-ups.
Although the exact cause of IBD remains unclear, researchers believe it results from a combination of genetic susceptibility, environmental triggers, immune system abnormalities, and lifestyle factors. Increasing urbanization, dietary changes, and stress levels have also been linked to rising incidence rates worldwide.
The long-term nature of IBD requires ongoing medical care, often involving a combination of medications, lifestyle adjustments, and in some cases surgical intervention. Advances in targeted therapies have dramatically improved patient outcomes over the last decade.
Key Growth Drivers of the IBD Treatment Market
Increasing Incidence of Inflammatory Bowel Diseases
One of the primary drivers of market growth is the rising prevalence of IBD across the globe. Changing lifestyles, dietary habits, and environmental factors have contributed to a noticeable increase in both Crohn’s disease and ulcerative colitis cases.
Globally, approximately 10 million people are currently living with inflammatory bowel disease. Data from a July 2023 study by the Crohn’s & Colitis Foundation revealed that around 721 cases per 100,000 people in the United States are diagnosed with IBD. In Canada, more than 320,000 individuals were estimated to have IBD in 2023, with projections suggesting this number could reach 470,000 by 2035.
The growing patient population is creating significant demand for advanced treatment options. As a result, pharmaceutical companies and research institutions are increasing investments in drug development and clinical trials.
Advancements in Biologic Therapies
Biologic therapies have revolutionized the treatment landscape for inflammatory bowel diseases. These medications target specific components of the immune system responsible for inflammation, offering more precise and effective treatment compared to traditional drugs.
Tumor necrosis factor (TNF) inhibitors and Janus kinase (JAK) inhibitors are among the most widely used biologic therapies. They have demonstrated strong clinical efficacy in reducing inflammation, preventing complications, and improving long-term disease remission.
In December 2023, Insilico Medicine began dosing the first patients with an investigational IBD drug candidate designed using generative artificial intelligence. Such innovations highlight how emerging technologies are accelerating pharmaceutical research and development.
As more biologic drugs enter the market, patients now have access to a broader range of targeted therapies that can significantly improve their quality of life.
Growing Awareness and Early Diagnosis
Awareness about inflammatory bowel disease has increased significantly in recent years. Public health campaigns, patient advocacy groups, and improved diagnostic tools have contributed to earlier detection and treatment.
Healthcare professionals are now better equipped to recognize symptoms and provide timely intervention. Early diagnosis helps prevent complications, improves treatment outcomes, and reduces the overall burden on healthcare systems.
Additionally, studies indicate a rising incidence of pediatric-onset IBD. According to estimates released by the U.S. National Institutes of Health in 2023, cases among children and adolescents are increasing, further emphasizing the need for improved therapeutic strategies.
Challenges in the Inflammatory Bowel Disease Treatment Market
High Cost of Advanced Therapies
Despite significant progress in treatment innovation, the high cost of biologic therapies remains a major barrier. These medications are often expensive due to complex manufacturing processes and research costs.
In many developing countries, limited healthcare coverage and affordability issues restrict patient access to these advanced treatments. Addressing this challenge requires policy initiatives, insurance support, and the development of cost-effective biosimilars.
Expanding access to affordable treatments will play a crucial role in ensuring equitable healthcare for IBD patients worldwide.
Side Effects and Limited Long-Term Effectiveness
Although biologic therapies are highly effective, they can also cause side effects in some patients. Increased susceptibility to infections and immune system complications are among the most common concerns.
Additionally, some individuals may develop resistance to certain medications over time, reducing their long-term effectiveness. As a result, physicians must often adjust treatment plans or combine therapies to maintain disease control.
Ongoing research into personalized medicine and new drug classes aims to address these limitations and provide safer, more durable treatment options.
Drug Class Insights in the IBD Treatment Market
TNF Inhibitors
TNF inhibitors remain one of the most widely used drug classes for managing inflammatory bowel diseases. These biologic drugs block tumor necrosis factor, a protein that plays a central role in inflammation.
Medications such as infliximab and adalimumab have demonstrated strong clinical effectiveness in reducing disease activity and maintaining remission. Their ability to improve patient outcomes has made them a cornerstone of modern IBD therapy.
As healthcare systems expand access to biologics, the demand for TNF inhibitors continues to grow worldwide.
JAK Inhibitors
JAK inhibitors represent a newer class of targeted therapies that interfere with the Janus kinase signaling pathway involved in immune responses.
Unlike many biologics that require injections or infusions, JAK inhibitors are typically administered orally. This convenience makes them an attractive option for patients who prefer non-injectable treatments.
One example is tofacitinib, which has shown promising results in patients who do not respond well to traditional biologic therapies. The continued development of small-molecule drugs is expected to expand treatment options in the coming years.
Disease Indication Insights
Ulcerative Colitis Treatment Market
The ulcerative colitis treatment segment represents a significant portion of the IBD therapy market. Rising disease prevalence and advancements in targeted drugs are contributing to increased demand for effective treatments.
Pharmaceutical companies are actively investing in research to develop new therapies that can reduce inflammation and maintain long-term remission. In April 2023, the European Commission approved Rinvoq (upadacitinib) for the treatment of moderate to severe Crohn’s disease in adults.
Clinical trials and collaborations between hospitals and pharmaceutical firms are accelerating innovation in ulcerative colitis treatment.
Crohn’s Disease Treatment Market
The treatment landscape for Crohn’s disease is evolving rapidly with the introduction of new biologics and targeted therapies. Researchers are focusing on therapies that address previously unmet medical needs.
Precision medicine approaches, dietary management strategies, and innovative biologic drugs are improving patient outcomes. Continued investments in clinical research are expected to drive major breakthroughs in Crohn’s disease management.
Distribution Channel Trends
Hospitals remain the primary treatment centers for inflammatory bowel disease. Their advanced diagnostic capabilities and multidisciplinary healthcare teams allow physicians to deliver comprehensive care.
Hospital pharmacies also play a crucial role in providing access to biologic therapies and specialized medications required for IBD management.
Meanwhile, the rise of telemedicine and digital health platforms is supporting the growth of online pharmacies. These services enable patients to receive consultations, prescription refills, and treatment guidance remotely, improving convenience and medication adherence.
Retail pharmacies also continue to serve as an important distribution channel, particularly for maintenance medications.
Regional Market Insights
United States
The United States represents one of the largest markets for inflammatory bowel disease treatments. High disease prevalence, advanced healthcare infrastructure, and strong pharmaceutical research activity contribute to market growth.
In January 2025, Eli Lilly announced that the U.S. Food and Drug Administration approved Omvoh (mirikizumab-mrkz) for the treatment of moderately to severely active Crohn’s disease in adults. The drug had previously received approval for ulcerative colitis in October 2023.
Such regulatory approvals demonstrate the rapid pace of innovation in the American pharmaceutical sector.
United Kingdom
The United Kingdom’s IBD treatment market is supported by a strong healthcare system and increasing awareness of autoimmune diseases.
The National Health Service continues to expand access to advanced therapies while supporting clinical research collaborations with pharmaceutical companies.
Recent estimates suggest that approximately half a million people in the UK were living with IBD in 2022, nearly double earlier estimates of 300,000 patients.
India
India’s IBD treatment market is witnessing steady growth due to rising disease prevalence and improved healthcare access.
Greater awareness among patients and healthcare professionals is encouraging early diagnosis and treatment. However, affordability challenges remain a concern, particularly in rural areas.
Healthcare providers are addressing this issue by expanding specialized treatment centers. In June 2023, Aster Medcity launched a dedicated Inflammatory Bowel Disease Centre to provide specialized care for patients.
Saudi Arabia
Saudi Arabia is also emerging as an important market for IBD treatment. The country’s growing healthcare investments and modern medical infrastructure are supporting improved disease management.
Government initiatives aimed at expanding healthcare access and adopting advanced therapies are expected to drive market expansion across the region.
Competitive Landscape
The inflammatory bowel disease treatment market is highly competitive, with several global pharmaceutical companies investing heavily in research and development. Leading players are focusing on developing innovative biologics, targeted therapies, and biosimilars to strengthen their market positions.
Key companies operating in the market include:
Abbott Laboratories
AbbVie Inc.
Allergan Therapeutics LLC
Bausch Health Companies Inc. (Salix Pharmaceuticals)
Bristol-Myers Squibb Company
Johnson & Johnson
Novartis AG
Pfizer Inc.
These companies are pursuing strategic collaborations, product launches, and clinical trials to expand their treatment portfolios and capture growing market demand.
Final Thoughts
Inflammatory bowel disease remains a complex chronic condition affecting millions of individuals worldwide. However, advances in biologic therapies, precision medicine, and digital healthcare are transforming how the disease is treated and managed.
With the global market expected to reach USD 33.99 billion by 2033, the future of IBD treatment appears promising. Continued investment in research, improved diagnostic capabilities, and better access to healthcare services will play a critical role in shaping the next generation of therapies.
Ultimately, the ongoing evolution of treatment strategies offers renewed hope for patients living with Crohn’s disease and ulcerative colitis, bringing the medical community closer to achieving long-term remission and improved quality of life for millions worldwide.
About the Creator
Sakshi Sharma
Content Writer with 7+ years of experience crafting SEO-driven blogs, web copy & research reports. Skilled in creating engaging, audience-focused content across diverse industries.





Comments
There are no comments for this story
Be the first to respond and start the conversation.