Pediatric Interventional Cardiology Market Size & Forecast 2026–2034
Rising Demand for Minimally Invasive Pediatric Heart Procedures to Drive Global Growth
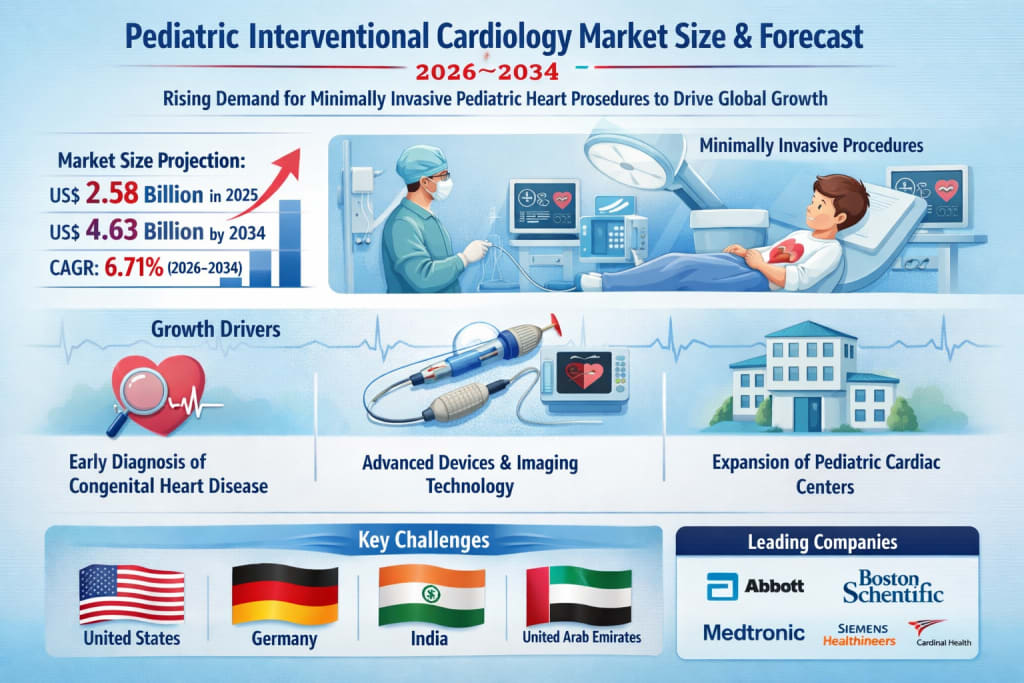
The global Pediatric Interventional Cardiology Market is entering a transformative growth phase. According to Renub Research, the market is projected to rise from US$ 2.58 billion in 2025 to US$ 4.63 billion by 2034, expanding at a CAGR of 6.71% from 2026 to 2034.
This steady expansion reflects more than just numbers. It highlights progress in pediatric healthcare, growing awareness of congenital heart disease (CHD), and rapid innovation in minimally invasive cardiac procedures. As healthcare systems prioritize safer, faster, and more effective treatments for children, pediatric interventional cardiology is emerging as a critical specialty within cardiovascular medicine.
Pediatric Interventional Cardiology Industry Overview
Pediatric interventional cardiology focuses on the diagnosis and catheter-based treatment of congenital and acquired heart conditions in infants, children, and adolescents. Unlike traditional open-heart surgery, these procedures use minimally invasive catheter techniques to correct structural abnormalities such as septal defects, valve disorders, and vascular blockages.
This approach significantly reduces recovery time, hospital stays, and procedural risks—factors that are especially important in pediatric populations.
The industry relies on highly specialized devices designed specifically for smaller anatomies and growing bodies. These include:
Catheters and guidewires
Balloons and stents
Occlusion devices
Transcatheter heart valves
Advanced imaging systems
Unlike adult cardiology, pediatric interventions must account for long-term growth and developmental considerations. Devices must balance flexibility, durability, and biocompatibility while enabling future adjustments or repeat interventions.
The field integrates advanced imaging technologies such as fluoroscopy, echocardiography, and 3D mapping systems, allowing physicians to perform complex procedures with high precision. Increasingly, hybrid procedures that combine surgical and catheter-based techniques are expanding treatment possibilities.
Hospitals, specialty pediatric cardiac centers, and academic institutions are the primary end users, supported by multidisciplinary teams of cardiologists, anesthesiologists, and pediatric intensivists.
Despite technological progress, the industry faces challenges such as limited pediatric-specific device portfolios, high R&D costs, and stringent regulatory approval pathways. Nevertheless, collaborative efforts between manufacturers and healthcare providers continue to accelerate innovation.
Growth Drivers in the Pediatric Interventional Cardiology Market
1. Increasing Prevalence and Early Diagnosis of Congenital Heart Disease
Congenital heart disease remains one of the most common birth defects globally. Advances in prenatal screening, fetal echocardiography, and neonatal diagnostics are enabling earlier detection of structural abnormalities.
Early diagnosis allows:
Timely intervention
Improved survival rates
Reduced complications
Increased eligibility for minimally invasive treatments
Greater awareness among parents and healthcare professionals is also leading to more frequent screenings and referrals to specialized pediatric cardiac centers.
Additionally, improvements in neonatal care have increased survival among premature infants and children with complex cardiac anomalies, expanding the patient population requiring interventional procedures.
As demand for safer, less invasive solutions grows, catheter-based treatments are increasingly preferred over traditional surgical approaches.
2. Technological Advancements in Devices and Imaging
Innovation is at the heart of this market’s growth. Manufacturers are developing devices specifically tailored for pediatric anatomies, including:
Growth-accommodating stents
Flexible occlusion devices
Miniaturized catheters
Implantable valves
Imaging technologies such as 3D echocardiography, hybrid catheterization labs, and advanced navigation systems enhance procedural accuracy and safety.
Robotics and real-time imaging guidance allow cardiologists to treat even high-risk and younger patients with improved outcomes. As technological sophistication increases, more hospitals adopt interventional procedures, further expanding market reach.
3. Expansion of Specialized Pediatric Cardiac Centers
Globally, healthcare systems are investing in dedicated pediatric cardiac units equipped with advanced catheterization laboratories. These centers provide comprehensive cardiac care under multidisciplinary supervision.
The presence of highly trained interventional cardiologists ensures:
High procedural success rates
Reduced complication risks
Better long-term patient outcomes
Medical training programs and specialized fellowships are also strengthening workforce expertise, particularly in emerging economies.
As more institutions adopt advanced interventional techniques, demand for pediatric cardiology devices and procedures continues to grow.
Challenges in the Pediatric Interventional Cardiology Market
Limited Pediatric-Specific Devices and High Development Costs
Unlike adult cardiology, pediatric cardiology serves a smaller and more diverse patient population. Developing devices suitable for small anatomies and long-term growth requires:
Extensive research
Complex engineering
Rigorous clinical testing
The smaller market size often limits return on investment, making innovation financially challenging.
Additionally, long clinical trial timelines and strict safety requirements increase development costs, potentially slowing the introduction of new technologies.
Regulatory and Reimbursement Complexities
Regulatory oversight for pediatric devices is stringent, particularly in major markets like the United States and Europe. Clinical studies involving children require robust safety data and ethical considerations, extending approval timelines.
Reimbursement variability across regions further complicates adoption. Novel minimally invasive procedures may face inconsistent insurance coverage, creating financial uncertainty for hospitals.
These challenges can limit patient access to advanced technologies despite their clinical benefits.
Regional Insights
United States Pediatric Interventional Cardiology Market
The U.S. market remains one of the most advanced globally, driven by strong clinical research, advanced imaging infrastructure, and innovation in pediatric devices.
Regulatory oversight from the U.S. Food and Drug Administration ensures high safety standards. The presence of leading medical device companies such as Abbott Laboratories, Boston Scientific Corporation, and Medtronic strengthens the innovation ecosystem.
Growing survival rates among children with complex heart defects and expansion of pediatric cardiac centers support steady growth.
Germany Pediatric Interventional Cardiology Market
Germany benefits from a robust healthcare system, strong neonatal screening programs, and advanced cardiac imaging capabilities.
Strict European regulatory standards ensure device quality, though they can slow approvals. Nonetheless, Germany remains a key European hub for pediatric cardiac research and innovation.
India Pediatric Interventional Cardiology Market
India represents a rapidly expanding market, driven by rising awareness of congenital heart disease and expanding healthcare infrastructure in major cities such as Mumbai, Delhi, and Bangalore.
Improved insurance penetration and government healthcare initiatives are supporting access to advanced cardiac treatments. However, high device costs and limited trained specialists in rural regions remain barriers.
United Arab Emirates Pediatric Interventional Cardiology Market
The UAE is emerging as a regional center for specialized pediatric cardiac care. Investments in healthcare infrastructure, advanced catheterization labs, and medical tourism are boosting growth.
While reliance on imported devices and limited local expertise present challenges, ongoing investments are positioning the UAE as a promising market.
Recent Industry Developments
The pediatric interventional cardiology market has witnessed several notable advancements:
June 2025: Canid secured $10 million in Series funding to enhance pediatric healthcare technology solutions.
April 2025: Abbott Laboratories received FDA approval for its Tendyne transcatheter mitral valve replacement system.
January 2025: Boston Scientific Corporation acquired Bolt Medical for up to USD 664 million to expand treatment capabilities for calcified artery disease.
December 2024: Johnson & Johnson obtained FDA clearance to expand indications for its Impella heart pump in younger patients.
These developments highlight growing investor confidence and technological momentum in pediatric cardiovascular innovation.
Market Segmentation
By Device Type
Transcatheter Heart Valves
Atherectomy Devices
Catheters
Closure Devices
Balloons
Stents
Others
By Procedure
Catheter-Based Valve Implantation
Congenital Heart Defect Correction
Angioplasty
Coronary Thrombectomy
Others
By End Use
Hospitals
Ambulatory Surgical Centers
Specialty Clinics
By Region
North America, Europe, Asia Pacific, Latin America, Middle East & Africa
Competitive Landscape
Key players shaping the global pediatric interventional cardiology market include:
Abbott Laboratories
Boston Scientific Corporation
General Electric
Medtronic
Siemens AG
Cardinal Health Inc.
These companies compete through product innovation, regulatory approvals, acquisitions, and strategic collaborations. SWOT analysis, revenue growth patterns, and pipeline developments remain key competitive indicators.
Final Thoughts
The Pediatric Interventional Cardiology Market stands at the intersection of innovation, compassion, and clinical precision. With projections indicating growth from US$ 2.58 billion in 2025 to US$ 4.63 billion by 2034, the market reflects expanding demand for minimally invasive pediatric cardiac solutions.
Driven by early diagnosis, technological breakthroughs, supportive healthcare policies, and expanding specialized centers, this sector is steadily transforming pediatric heart care worldwide.
While regulatory hurdles and high development costs present ongoing challenges, continuous collaboration among medical device manufacturers, clinicians, and policymakers ensures sustained progress.
As innovation advances and awareness grows, pediatric interventional cardiology will continue to improve survival rates, enhance quality of life, and shape the future of cardiac care for the youngest and most vulnerable patients.






Comments
There are no comments for this story
Be the first to respond and start the conversation.